Our Research Focus
Diseases due to Protein Aggregation, Protein Misfolding & Neurodegeneration
Introduction to the research field:
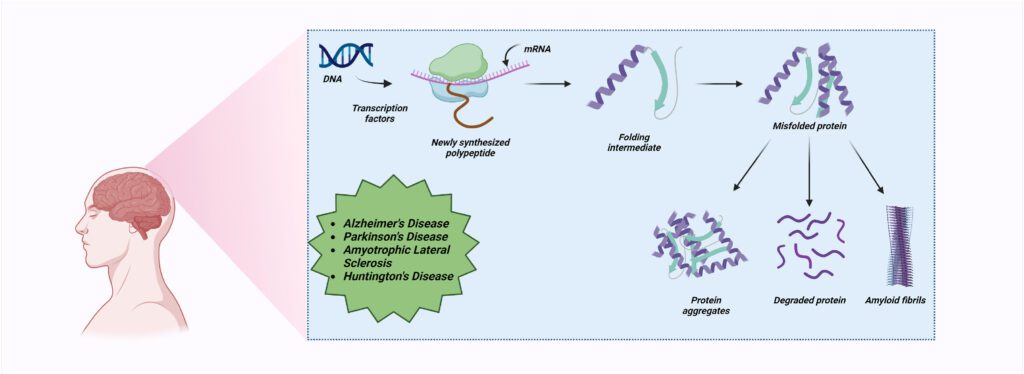
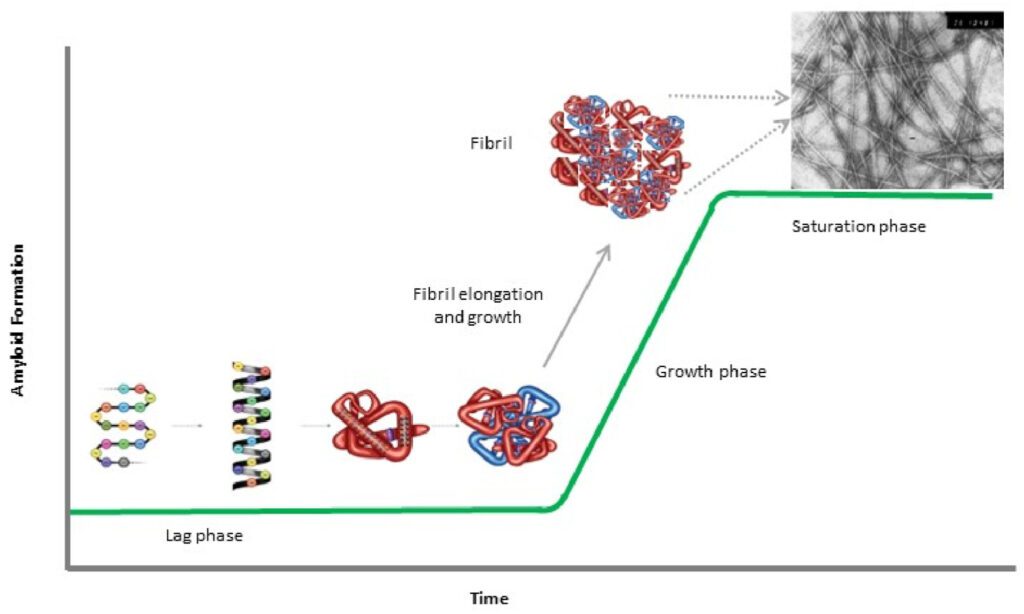
Protein misfolding and aggregation are key events in the pathogenesis of several neurodegenerative diseases. Proteins, which are crucial for the functioning of cells, are made up of amino acids and fold into specific three-dimensional structures. This folding process is essential for their function. However, errors in this process can lead to the formation of incorrectly folded proteins, which can aggregate and cause diseases. One of the most well-known diseases associated with protein misfolding and aggregation is Alzheimer’s disease. In this condition, the protein amyloid-beta misfolds and aggregates to form plaques in the brain, leading to neuronal damage and the symptoms of dementia. Similarly, in Parkinson’s disease, the protein alpha-synuclein misfolds and forms aggregates known as Lewy bodies, which cause the death of dopamine-producing neurons and result in the motor symptoms of the disease. Another example is Huntington’s disease, which is caused by the aggregation of the huntingtin protein. This leads to the death of neurons in certain areas of the brain, resulting in movement, cognitive, and psychiatric symptoms. Creutzfeldt-Jakob disease, a rare and fatal condition, is also associated with protein misfolding and aggregation. In this case, prion proteins misfold and aggregate, causing rapid and severe neurodegeneration. In addition to these, there are several other diseases associated with protein misfolding and aggregation, including Amyotrophic Lateral Sclerosis (ALS), Frontotemporal Dementia (FTD), and certain forms of spinocerebellar ataxia. These conditions, while diverse in their symptoms and progression, all share the common feature of protein misfolding and aggregation leading to neuronal damage and death. It’s important to note that the relationship between protein misfolding, aggregation, and disease is complex. While aggregation of misfolded proteins is clearly associated with disease, it’s not always clear whether the aggregates themselves are causing damage, or whether they are a byproduct of other cellular processes gone awry. Furthermore, the exact mechanisms by which misfolded proteins lead to neuronal death are still not fully understood and are the subject of ongoing research. In conclusion, protein misfolding and aggregation are central to the pathogenesis of several neurodegenerative diseases. Understanding these processes and finding ways to prevent or reverse them is a major focus of research in the field of neurodegeneration. It is hoped that this research will eventually lead to effective treatments for these currently incurable conditions.
Focus:
We work with crowders to find out solutions that interferes with normal activity of proteins.
Publication:
Below are some list of publications on this topic.
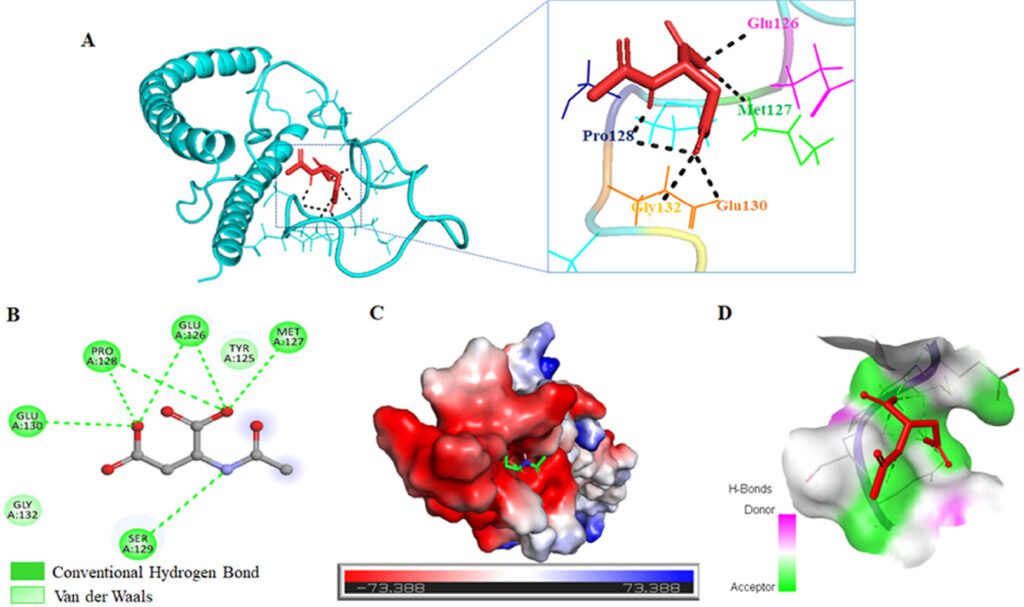
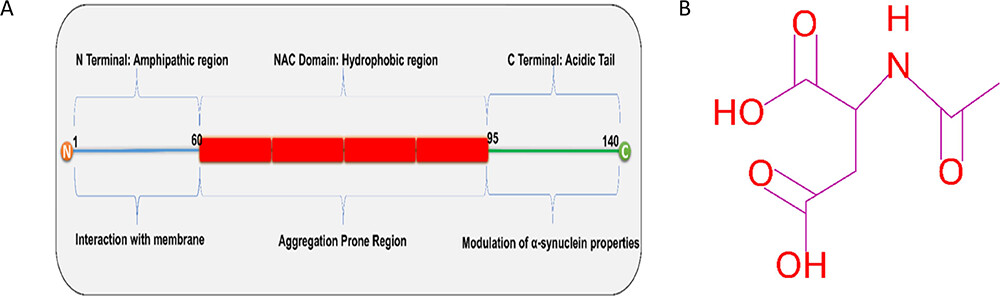
Understanding the Modulation of α-Synuclein Fibrillation by N-Acetyl Aspartate: A Brain Metabolite
Recent advancement in therapeutic strategies for Alzheimer’s disease: Insights from clinical trials
- Navigating the Maze of Alzheimer’s disease by exploring BACE1: Discovery, current scenario, and future prospects
Protein Folding and Dynamics
Introduction to the research field:
Protein folding and dynamics is a fascinating field of study in molecular biology and biochemistry. It involves the process by which a protein structure, a specific sequence of amino acids, folds into its functional three-dimensional structure. This process is critical to many aspects of protein function and regulation. Proteins are the workhorses of the cell, performing a vast array of functions. The function of a protein is directly related to its 3D structure. Amazingly, this structure is determined solely by the protein’s amino acid sequence. But how does a protein’s amino acid sequence direct it to fold into its correct structure? This is the fundamental question in the field of protein folding. The folding and unfolding of proteins is a dynamic process. When we think about how proteins fold, we have to think dynamically as well as thermodynamically. Correct folding allows a polypeptide chain to adopt its functional three-dimensional structure. However, improper folding can lead to the aggregation of misfolded proteins, which is linked to many human diseases. The process of protein folding is not fully understood. It is known that the protein folding process is cooperative, meaning that the formation of one structure helps to form others. Protein folding is a stochastic process and proteins unfold and refold in a dynamic equilibrium. The effect of temperature on the stability of the folded and unfolded states is also a key factor. Molecular dynamics (MD) tools help us imagine how these processes take place and concomitantly how to probe protein folding experimentally. As protein folding occurs in 3D, let’s explore a free energy (G) landscape for folding from an extraordinarily large number of unfolded states of higher free energy to a single low energy folded state. In the field of protein folding and dynamics, researchers use a variety of experimental and computational techniques to study these processes. These techniques help to validate, simulate, predict, and analyze protein structures. The knowledge gained from these studies has applications in various fields, including drug design, disease prediction, and the engineering of proteins with novel properties. In conclusion, protein folding and dynamics is a complex and intriguing field that combines elements of biology, chemistry, physics, and computer science. It is a field that has the potential to provide insights into the fundamental processes of life, as well as to contribute to the development of new therapies for a range of diseases.
Focus:
We work with crowders to find out solutions that interferes with normal activity of proteins.
Publication:
Below are some list of publications on this topic.
Understanding the Modulation of α-Synuclein Fibrillation by N-Acetyl Aspartate: A Brain Metabolite
Macromolecular Crowding
Inrtroduction to the Research Field:
Macromolecular crowding is a phenomenon that occurs in the cytosol of cells where the properties of macromolecules such as proteins and nucleic acids are altered due to high concentrations of these macromolecules. This condition is a result of the high density of intracellular macromolecules which creates a special environment known as macromolecular crowding. The phenomenon of macromolecular crowding alters the properties of molecules in a solution when high concentrations of macromolecules such as proteins are present. Such conditions occur routinely in living cells; for instance, the cytosol of Escherichia coli contains about 300–400 mg/ml of macromolecules. Crowding occurs since these high concentrations of macromolecules reduce the volume of solvent available for other molecules in the solution, which has the result of increasing their effective concentrations. Crowding can promote the formation of a biomolecular condensate by colloidal phase separation. This crowding effect can make molecules in cells behave in radically different ways than in test-tube assays. Consequently, measurements of the properties of enzymes or processes in metabolism that are made in the laboratory (in vitro) in dilute solutions may be different by many orders of magnitude from the true values seen in living cells (in vivo). The study of biochemical processes under realistically crowded conditions is very important, since these conditions are a ubiquitous property of all cells and crowding may be essential for the efficient operation of metabolism. Indeed, in vitro studies have shown that crowding greatly influences the binding stability of proteins to DNA. The interior of cells is a crowded environment. For example, an Escherichia coli cell is only about 2 micrometres (μm) long and 0.5 μm in diameter, with a cell volume of 0.6 – 0.7 μm 3. However, E. coli can contain up to 4,288 different types of proteins, and about 1,000 of these types are produced at a high enough level to be easily detected. Added to this mix are various forms of RNA and the cell’s DNA chromosome, giving a total concentration of macromolecules of between 300 and 400 mg/ml. In eukaryotes the cell’s interior is further crowded by the protein filaments that make up the cytoskeleton, this meshwork divides the cytosol into a network of narrow pores. The volume of accessible solvent (red) for two molecules of widely different sizes (black circles) at high concentrations of macromolecules (grey circles). Reducing the available volume increases the effective concentration of macromolecules. These high concentrations of macromolecules occupy a large proportion of the volume of the cell, which reduces the volume of solvent that is available for other macromolecules. This excluded volume effect increases the effective concentration of macromolecules (increasing their chemical activity), which in turn alters the rates and equilibrium constants of their reactions. In particular this effect alters dissociation constants by favoring the association of macromolecules, such as when multiple proteins come together to form protein complexes, or when DNA-binding proteins bind to their targets in the genome.
Focus:
We work with crowders to find out solutions that interferes with normal activity of proteins.
Publication:
Below are some list of publications on this topic.
Understanding the Modulation of α-Synuclein Fibrillation by N-Acetyl Aspartate: A Brain Metabolite
Co-solute Engineering
Introduction to the research field:
Co-solute engineering is a fascinating field that deals with the manipulation and control of solute properties in a solution to achieve desired outcomes. This field is particularly relevant in the context of biochemical and chemical processes where the behavior of solutes can significantly impact the efficiency and effectiveness of these processes. In essence, co-solute engineering involves the use of co-solvents or co-solutes to modify the properties of a primary solute in a solution. This can be achieved by altering the solubility, reactivity, stability, and other physicochemical properties of the solute. The choice of co-solvent or co-solute is critical and depends on the specific requirements of the process. For instance, in the pharmaceutical industry, co-solute engineering is often employed to enhance the solubility of poorly soluble drugs. This is achieved by using co-solvents or complexation agents that can interact with the drug molecules and increase their solubility in the formulation. This not only improves the bioavailability of the drug but also allows for more efficient drug delivery. Similarly, in the field of biotechnology, co-solute engineering can be used to control the behavior of proteins in solution. By carefully selecting co-solutes, it is possible to influence protein folding, stability, and aggregation behavior. This can be particularly useful in the production of therapeutic proteins where maintaining the correct protein conformation is crucial for their biological activity. Moreover, co-solute engineering can also play a significant role in the field of materials science. For example, in the synthesis of nanoparticles, the use of appropriate co-solutes can help control the size, shape, and distribution of the nanoparticles. This can greatly influence the properties of the resulting material and its suitability for various applications. In conclusion, co-solute engineering is a powerful tool that allows scientists and engineers to manipulate the properties of solutes in a solution. By carefully selecting and controlling the co-solutes, it is possible to tailor the behavior of the primary solute to meet specific requirements. This field has wide-ranging applications in various industries including pharmaceuticals, biotechnology, and materials science, and continues to be an area of active research and development.
Focus:
We work with crowders to find out solutions that interferes with normal activity of proteins.
Publication:
Below are some list of publications on this topic.
Understanding the Modulation of α-Synuclein Fibrillation by N-Acetyl Aspartate: A Brain Metabolite
Bioremediation
Introduction to the research field:
Bioremediation is a fascinating and increasingly important field of study in environmental science. It refers to the use of living organisms, primarily microorganisms, to degrade the environmental contaminants into less toxic forms. These organisms, such as bacteria, fungi, and plants, have the natural ability to break down various harmful substances and transform them into harmless products. The process of bioremediation can be applied to a wide range of pollutants including oil, heavy metals, pesticides, and other organic compounds. The organisms used in bioremediation processes utilize these pollutants as a source of energy and nutrients, effectively cleaning up the contaminated environment while they grow and multiply. There are two main types of bioremediation: in situ and ex situ. In situ bioremediation involves treating the contaminated material at the site, while ex situ involves the removal of the contaminated material to be treated elsewhere. Different methods of bioremediation are used depending on the site and its contaminants. In addition to being a natural process, bioremediation has several advantages over traditional methods of pollution cleanup. It is often less expensive, more environmentally friendly, and can be used to treat a wide variety of contaminants. Furthermore, bioremediation can often be carried out on site, avoiding the need to transport contaminated material. However, bioremediation is not a one-size-fits-all solution. The effectiveness of bioremediation can be influenced by various factors including the type and concentration of the contaminant, the presence of suitable microorganisms, and environmental conditions such as temperature, pH, and the presence of oxygen. Despite these challenges, advances in biotechnology are making it possible to enhance the effectiveness of bioremediation. For example, through genetic engineering, scientists are now able to create organisms that are specifically designed to degrade certain pollutants. In conclusion, bioremediation represents a promising and environmentally friendly approach to managing pollution. As research in this field continues to advance, it is likely that we will see the development of more effective and versatile bioremediation strategies in the future. This not only has implications for environmental protection but also for industries such as waste management and agriculture. Bioremediation, therefore, holds the potential to significantly contribute to sustainable development and a cleaner, healthier environment.
Focus:
We work with crowders to find out solutions that interferes with normal activity of proteins.
Publication:
Below are some list of publications on this topic.
Understanding the Modulation of α-Synuclein Fibrillation by N-Acetyl Aspartate: A Brain Metabolite
- Navigating the Maze of Alzheimer’s disease by exploring BACE1: Discovery, current scenario, and future prospects
- Recent advancement in therapeutic strategies for Alzheimer’s disease: Insights from clinical trials